Hepatitis E Virus
With an estimated 20 million infections per year, HEV is one of the most common causes of acute viral hepatitis worldwide. Although infections usually resolve spontaneously, they can become chronic in immunocompromised individuals. This can lead to extrahepatic manifestations as well as cirrhosis and liver failure. In addition, an increased risk of miscarriage and a mortality rate of up to 30% have been observed in pregnant women infected with a particular HEV genotype. There is currently no specific therapy against HEV and the broad-spectrum antiviral agent ribavirin is the only treatment option. However, this can lead to development of resistance and treatment failure, which is associated with mutations in the viral polymerase.
Our HEV research mainly focuses on three areas:
1. Mechanisms of HEV replication, resistance and chronic persistence.
One aim of our research is to investigate genetic determinants of HEV polymerase before and during ribavirin therapy to better understand its mode of action. Using deep sequencing, we rencently demonstrated that ribavirin exerts a mutagenic effect on the viral genome, which may lead to viral elimination and/or selection of resistant variants (Todt et al., 2016). This approach can be used in the future to identify patients at risk of treatment failure.
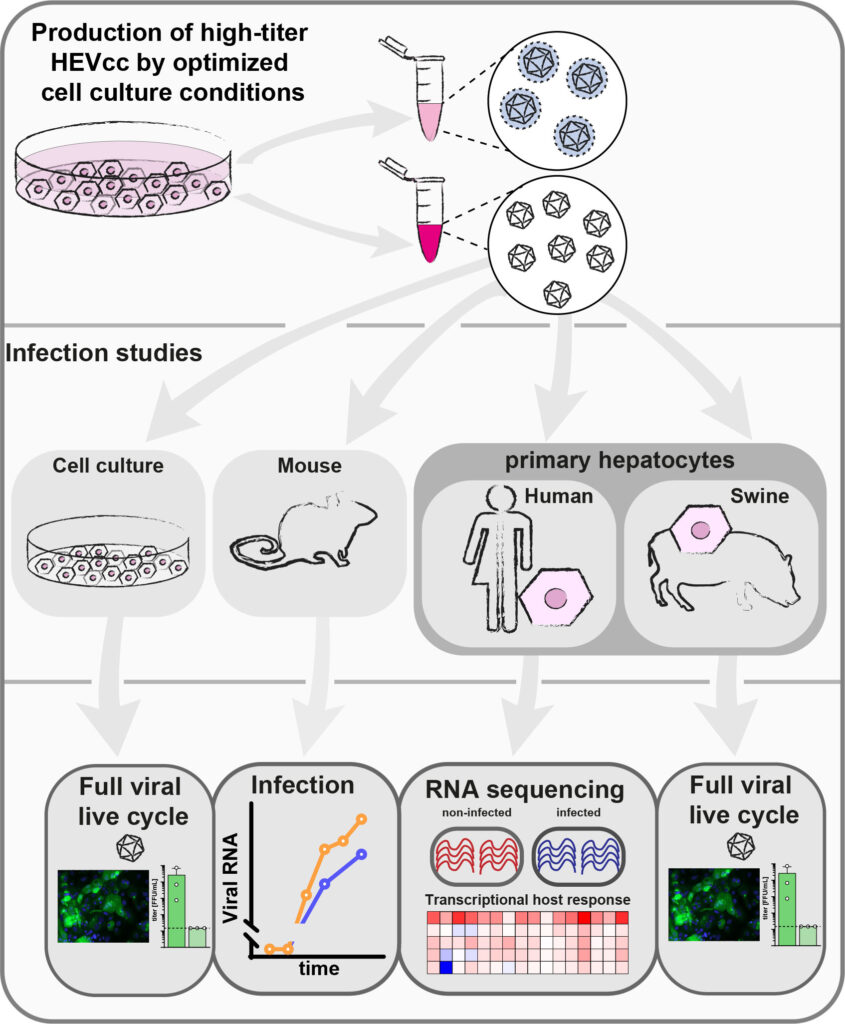
Furthermore, we analyze mechanisms of HEV replication, using a recently established novel cell culture system (Todt et al., 2020). We aim to identify host factors required for HEV replication and thereby reveal potential novel drug targets.
2. Identification and characterization of novel antiviral agents.
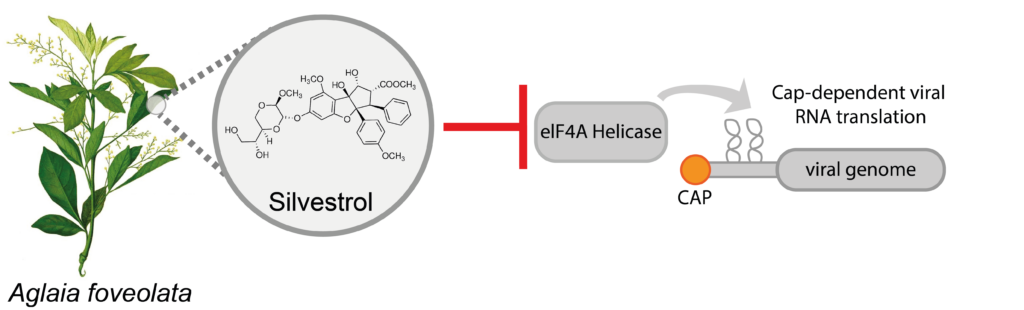
In order to improve the treatment of HEV patients, we are searching for new antiviral substances and therapy options. Since interferon-alpha has been tested in individual cases for the treatment of HEV infections, we have analyzed the effect of different interferons against HEV (Todt et al., 2016). Recently, we also demonstrated that the natural compound silvestrol inhibits HEV replication and production of new viruses (Todt and Moeller et al., 2018). In the future, we hope to find more antiviral agents and also help to transfer our discoveries towards clinical applications.
3. Development of HEV cell culture models
The lack of efficient cell culture models is one of the main reasons why many questions about HEV infection biology have not been resolved so far. Recently, we have established a simple but robust cell culture model for HEV that allows to produce more than 100-fold infectious virus particles compared to existing model systems (Todt et al., 2020). The obtained virus particles allow to recapitulate the entire life cycle of HEV in vitro. Using our cell culture system, we are able show that HEV infections are not restricted to liver cells and HEV can also complete its life cycle in neuronal and in placental cells (Drave et al., 2016 ; Knegendorf et al., 2018). In addition, we are using our cell culture system to study basic HEV infection biology and test novel antiviral drug candidates.
Our Team:

Eike Steinmann
Professor
Yannick Brüggemann
PhD
Michelle Jagst
PhD Candidate
Sarah Schlienkamp
PhD Candidate